


 |  |  | 13.4 Release mechanisms of phosphorus from sediment to water |
After settling, particulate P begins to undergo chemical and biological reactions that will lead to dissolution of P (Figure 3). Two mechanisms have to occur simultaneously or within a short period before P is released from sediment to water: first, dissolution of P bound to particles or aggregates and its transfer to the pool of dissolved P in the pore water and, second, a transport process responsible for the release of dissolved P from sediment to water [56]. In sediments, P bound to particles is mobilized into dissolved form in desorption, dissolution, ligand exchange and enzymatic hydrolysis. The driving force behind the released P may be the rate at which dissolved P is generated in the pore water by splitting mechanisms of particulate P. In sediment, the dissolved P generated can be adsorbed, complexed with (in)organic cations, precipitated as authigenic minerals, and taken up by bacteria and benthic microalgae before being released to water.
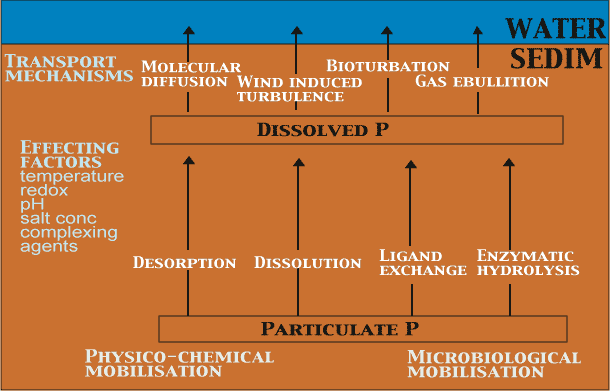
The main physical factors enhancing the exchange of P between sediment and water are molecular diffusion, temperature, water turbulence, gas ebullition and bioturbation. In stagnant anoxic hypolimnetic and non-bioturbated sediments, molecular diffusion is considered to be the main mechanism for P release. There is usually a pronounced difference in P concentrations between the overlying water and the surface pore water. The steeper the concentration gradient of P between the free water and the sediment pore water, the higher is the rate of diffusion. An increase in sediment solid matter content decreases free diffusion owing to the increase in collisions of molecules with the particles in sediments.
Temperature can have a major effect on the release rate of P from sediment to water. The changes in temperature affects on the microbially and chemically mediated redox reactions in sediments. An increase in temperature accelerates also the diffusion rate as a result of increased Brownian movement. However, temperature also has an indirect effect on P release, because an increase in temperature raises the metabolic rate of bacteria and subsequently the mineralization rate of organic matter.
In addition to molecular diffusion physical water movement can transport solutes from sediment to water. Physical water movement is a much faster transport mechanism than diffusion. Advective flux denotes transport of dissolved chemical species due to transport of water by water turbulence at the sediment-water interface, gas convection, seepage and organisms. The measured solute flux, for example by incubations, is usually higher than the calculated diffusion flux. Physical pore water movement is increased by water currents at the sediment-water interface and by gases formed in microbial reduction processes in sediments. In addition, bioturbation promotes physical water movement and affects the binding capacity of P in sediments.
Sediments are often inhabited by abundant and diverse populations of burrowing macroinverte-brates; these influence nutrient binding and release. A significant variation exists in the behaviour of different burrowing macroinvertebrate species, and so the effect of solute transport in and out of the sediments may vary considerably between species [310]. The most common mechanism by which they promote sediment-water exchange is, however, thought to be physical: through their pumping movements (digging and irrigation) in burrows the animals increase the contact between interstitial water and the overlying water. Further, benthic macroinvertebrates can alter sediment properties and chemical exchange between sediment and water by their respiration, feeding and defecating activities.
Burrowing has a three-way effect on the cycling of P in sediments: first, it increases the concentration of O2 in the burrows, thereby improving the P-binding capacity of sediment, but it also promotes the transport of pore water P from sediment to water; second, an abundance of macroinvertebrates increases the rate of mineralization [211], or the acceleration of their digestive and excretory processes [169] furthers the dissolution of P; and third, burrowing animals move particulate oxidized compounds down to the reduced zone and reduced compounds up to the oxic zone, thus affecting Fe(III) oxide reduction and re-oxidation in sediments [70].
 |  |  | 13.4 Release mechanisms of phosphorus from sediment to water |