


 |  |  | 13.2 Significance of benthic phosphorus flux |
Sediments are considered to act as `sinks' for particulate P. However, a portion of the P is released back into the water as a result of the biogeochemical processes undergone by settled particulate P during burial. The released P may significantly increase the biologically available pool of P in water, counteracting decreases in the external load. Owing to P released from the bottom, the total external load of P does not correspond fully to the pool of algal-available P in water. Thus, reducing the external load of P does not necessarily lead to rapid recovery of the system. In eutrophic lakes and the Baltic Sea, the benthic flux of P can temporarily equal or even exceed the input of P from external sources [100][55] and can delay recovery of these systems. The P cycle in sediments may take several decades to adjust to changing trophic conditions. In addition, one has to keep in mind that P released from sediment to water is almost completely in the form of dissolved reactive P (ortho-phosphate), which is, in turn, considered to be completely available for algae. A large part of the external P load enters to water systems as particulate P, which availability to algae is poor. Thus, the algal availability in the load of total P from external sources is not comparable to the efflux from the sediment to water.
In terms of P in sediments, two issues are related to the fate of P: the fraction of settled P that will eventually be buried in the sediments, and the fraction of P that will be transformed into an available form, i.e. one that may potentially contribute to eutrophication, before permanent burial. When P enters the aquatic system, internal processes affect the cycling of P in both water and sediment. In sediments some of the internal processes increase but some decrease the pool of bioavailable P in water. The benthic P efflux, referring to a commonly used term for the internal P load, originates from bottom sediments when dissolved P is released from particles to the pore water and subsequently to the overlying free water. Note that the benthic P efflux can only be based on P transported from external sources. The present section concentrates on the release of dissolved P and thus does not cover the effect of resuspension (i.e. transport of settled particles rich in P back to the water column).
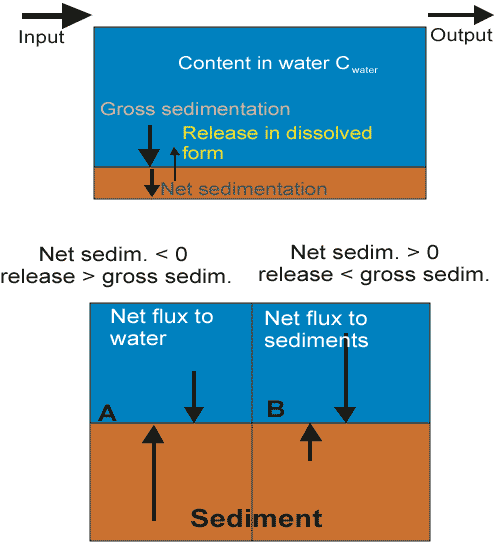
Sediments can, however, bind and release P. A net flux of P into sediments occurs when net sedimentation of P is above zero and P release is lower than gross P sedimentation (Figure 1). A net flux of P from sediment to water, in turn, occurs when net sedimentation of P is below zero and release is higher than gross P sedimentation. The flux in and out of the sediments may vary seasonally as well as between years, depending, among other things, on hydrological features. The retention of P in sediments is considered to be largely dependent on (1) net sedimentation of particulate P, (2) early diagenetic cycling and transformations of reactive P compounds, and (3) burial of immobilizable P compounds in deeper sediment layers [50].
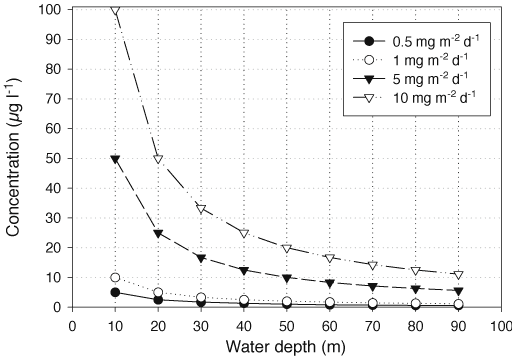
The release of P from sediment to water can be quantified with mass balance analysis at the whole-system level (mainly lakes), laboratory or in situ confinement of sediments in core tubes or benthic chambers, and also by diffusion flux calculations based on measured concentration gradients at the sediment-water interface. The increase in the concentration of dissolved substances in water after their release is controlled by the flux rate of the substances from sediment to water and the volume of water assuming that no back precipitation occurs. In a shallow area, for example, where the water depth is 10 m, an average flux rate of 1 mg m-2 d-1 during the summer period (100 d) increases the concentration of dissolved substances in the whole water column above this square metre to 10 µg l-1 after complete mixing (Figure 2). A similar increase in concentration is achieved at an average flux rate of 5 mg m-2 d-1 for water at a depth of 50 m.
In the case of nutrients, the issue of whether the benthic flux is high or low depends on the consequences of the efflux related to primary production. For example, the bioavailable P released from the sediments contributes to new production, i.e. is a fraction of total primary production. In relatively shallow aquatic environments such as estuaries, continental shelves and bays, the P efflux may support a large proportion of total primary production. In the upper Potomac River estuary, for example, efflux of P can meet 27% of the gross primary production requirements [67]. In Narrangansett Bay, efflux of P contributes about 50% (Nixon 1981), in the Kiel Bight 20% [32] and in Århus Bay, Denmark, 15% [250] of the amount needed to support primary production.
Especially in the eutrophic non-stratified shallow lakes the efflux of nutrients from sediment to their rather small water volume may be crucial during productive season. In these systems the nutrients in near-bottom water are rapidly transported to the euphotic water layer during water mixing.
Søndergaard et al. [436] have reported very high P flux rates up to 200 mg m-2 d-1 in hyper-eutrophic lake Søbygård (Denmark). These kind of high benthic release rates can be measured only temporarily, but in comparison, the release rate of 200 mg P m-2 d-1 per one km2 correspond daily total phosphorus load of about 67 000 persons, assuming that the total P load of one person per day is 3 g.
Sediment can, however, retain P well. In oligotrophic lakes the release of P from sediment to water is often almost undetectable in oxic conditions and low temperatures in short sediment incubations. For example, in oligotrophic lakes such as Lake Michigan the mean release rate of P 0.05 mg m-2 d-1 can supply less than 1% of the P utilized for annual primary production [102]. In addition, agriculturally P loaded Lake Pyhäjärvi in Finland retains in lake -- including fish catch -- almost 90% of the external total P load [148].
 |  |  | 13.2 Significance of benthic phosphorus flux |